Hydrolyzed Collagen
Help on accessing alternative formats, such as Portable Document Format (PDF), Microsoft Word and PowerPoint (PPT) files, can be obtained in the alternate format help section.
This monograph is intended to serve as a guide to industry for the preparation of Product Licence Applications (PLAs) and labels for natural health product market authorization. It is not intended to be a comprehensive review of the medicinal ingredient.
Notes- For the purpose of this monograph, hydrolyzed collagen has no jelling power and is soluble in cold water (Schrieber and Gareis 2007; Moskowitz 2000).The average molecular weight of hydrolyzed collagen is approximately 4 kDa (i.e. 2-6 kDa) (Moskowitz 2000; Oesser et al. 1999).
- Text in parentheses is additional optional information which can be included on the PLA and product label at the applicant's discretion.
- The solidus (/) indicates that the terms and/or statements are synonymous. Either term or statement may be selected by the applicant.
Date
March 28, 2024
Proper name(s), Common name(s), Source information
| Proper name(s) | Common name(s) | Source information | |
|---|---|---|---|
| Source material(s) | Part(s) | ||
| Hydrolyzed collagen |
|
Bovine | Bovine skin/hide split |
| Porcine |
|
||
| Fish |
|
||
| Chicken | Cartilage | ||
References: Proper name: NIH 2023, ICIDH 2008; Common names: NIH 2023, ICIDH 2008, Moskowitz 2000; Source information: Schauss et al 2012, FCC 7 2010, Schrieber and Gareis 2007, Baziwane and He 2003.
Route of administration
Oral
Dosage form(s)
This monograph excludes foods or food-like dosage forms as indicated in the Compendium of Monographs Guidance Document.
Acceptable dosage forms for oral use are indicated in the dosage form drop-down list of the web-based Product Licence Application form for Compendial applications.
Use(s) or Purpose(s)
- Source of (the) essential amino acid(s) (histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, valine) for the maintenance of good health/involved in (muscle) protein synthesis (CNF 2023; IOM 2005; Eastoe 1955).
- Source of (the) non-essential amino acid(s) (alanine, arginine, aspartic acid, glutamic acid, glycine, proline, serine, tyrosine) involved in (muscle) protein synthesis (CNF 2023; IOM 2005; Eastoe 1955).
- Source of (the essential amino acid) lysine to help in collagen formation/synthesis (derMarderosian and Beutler 2011; Baziwane and He 2003; Garrison and Somer 1995; Jansen 1962).
- Helps reduce joint pain associated with osteoarthritis (Bruyère et al. 2012; Benito-Ruiz et al. 2009; Clark et al. 2008).
- Helps reduce osteoarthritis-related joint pain (Bruyère et al. 2012; Benito-Ruiz et al. 2009; Clark et al. 2008).
- Helps manage/in the management of joint pain (Bruyère et al. 2012; Benito-Ruiz et al. 2009; Clark et al. 2008).
Notes:
- The above uses can be combined on the product label (e.g. Source of essential amino acids for the maintenance of good health and non-essential amino acids involved in muscle protein synthesis).
- The terms 'Helps' or 'Helps to' can be used interchangeably on the label.
Dose(s)
Subpopulation(s)
Adults 18 years and older
Quantity(ies)
Source of (the) essential amino acid(s)(histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, valine)
2.6-10 grams hydrolyzed collagen, per day (IOM 2005; Moskowitz 2000).
OR
| Essential amino acids |
Minimum dose (mg/day) 5% of the RDA1 |
Maximum dose of hydrolyzed collagen (g/day)2 |
|---|---|---|
| L-Histidine | 49 mg | 10 g |
| L-Isoleucine | 66.5 mg | |
| L-Leucine | 147 mg | |
| L-Lysine | 133 mg | |
| L-Methionine | 66.5 mg | |
| L-Phenylalanine | 115.5 mg | |
| L-Threonine | 70 mg | |
| L-Valine | 84 mg |
1Minimum doses have been calculated as 5% of each specific amino acid Recommended Dietary
Allowance with a reference weight of 70 kg (IOM 2005).
2Maximum dose (Benito-Ruiz et al.
2009;
IOM 2005; Moskowitz 2000).
Source of (the) non-essential amino acid(s) (alanine, arginine, aspartic acid, glutamic acid, glycine, proline, serine, tyrosine)
2.6-10 grams hydrolyzed collagen, per day (IOM 2005; Moskowitz 2000).
OR
| Non-Essential amino acids |
Minimum dose (mg/day) 5% of the RDA1 |
Maximum dose of hydrolyzed collagen (g/day)2 |
|---|---|---|
| L-Alanine | 181.5 mg | 10 g |
| L-Arginine | 208.5 mg | |
| L-Aspartic acid | 325 mg | |
| L-Glutamic acid | 750 mg | |
| Glycine | 160 mg | |
| L-Proline | 259.5 mg | |
| L-Serine | 175.5 mg | |
| L-Tyrosine | 139 mg |
1Minimum doses have been calculated as 5% of each specific amino acid Mean Intake (IOM
2005)
2Maximum dose (Benito-Ruiz et al. 2009; IOM 2005; Moskowitz 2000).
Source of lysine
| Essential amino acids |
Minimum dose(mg/day) 5% of the RDA1 |
Maximum dose of hydrolyzed collagen (g/day)2 |
|---|---|---|
| L-Lysine | 133 mg | 10 g |
1Minimum doses have been calculated as 5% of Recommended Dietary Allowance with a reference
weight of 70
kg (IOM 2005).
2Maximum dose (Benito-Ruiz et al. 2009; IOM 2005; Moskowitz 2000).
Joint pain
1.2-10 grams hydrolyzed collagen, per day (Bruyère et al. 2012; Benito-Ruiz et al. 2009; Clark et al. 2008).
Direction(s) for use
No statement required.Duration(s) of use
Joint pain
Use for at least 5 months to see beneficial effects (Bruyère et al. 2012; Benito-Ruiz et al. 2009; Clark et al. 2008).Risk information
Caution(s) and warning(s)
Joint pain
Ask a health care practitioner/health care provider/health care professional/doctor/physician if symptoms worsen.
Products providing more than 2.8 g hydrolyzed collagen, per day
Ask a health care practitioner/health care provider/health care professional/doctor/physician before use if you are pregnant or breastfeeding (Shils et al. 2006; Goldman and Ausiello 2004).
Contraindication(s)
No statement required.
Known adverse reaction(s)
When using this product you may experience gastrointestinal discomfort/disturbances (Moskowitz 2000).
Non-medicinal ingredients
Must be chosen from the current Natural Health Products Ingredients Database and must meet the limitations outlined in the database.
Storage conditions
Must be established in accordance with the requirements described in the Natural Health Products Regulations.
All products (information for industry; optional for labelling depending on the packaging)
To be protected from heat and moisture (Ph.Eur. 2023).
Specifications
- The finished product specifications must be established in accordance with the requirements described in the Natural and Non-Prescription Health Products (NNHPD) Quality of Natural Health Products Guide.
- The medicinal ingredient must comply with the requirements outlined in the NHPID .
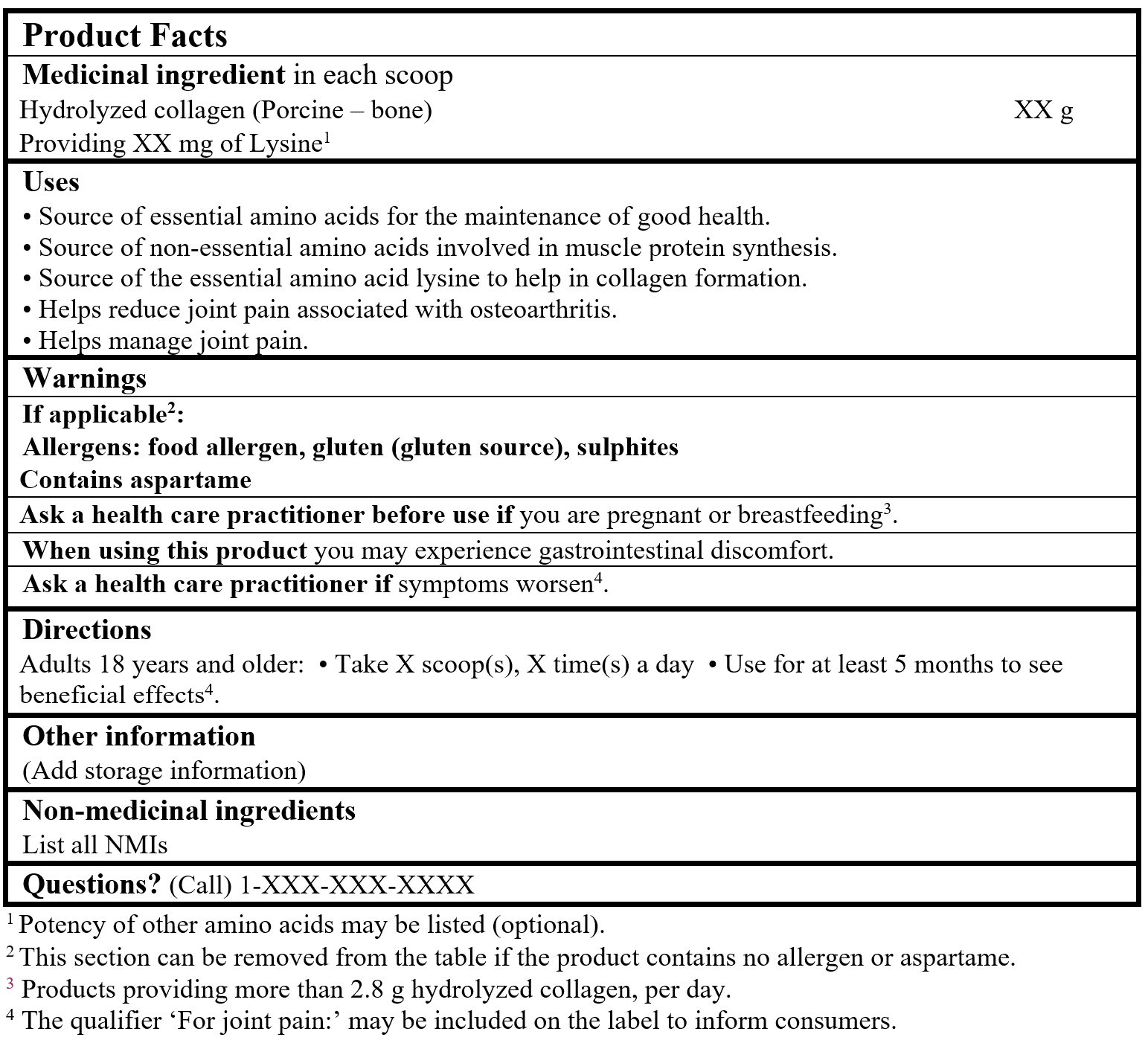
EXAMPLE OF PRODUCT FACTS:
Consult the Guidance Document, Labelling of Natural Health Products for more details.
References cited
- Baziwane D, He Q. Gelatin: The paramount food additive. Food Reviews International 2003;19(4):423-435.
- Benito-Ruiz P, Camacho-Zambrano MM, Carrillo-Arcentales JN, Mestanza-Peralta MA, Vallejo-Flores CA, Vargas-López SV, Villacís-Tamayo RA, Zurita-Gavilanes LA. A randomized controlled trial on the efficacy and safety of a food ingredient, collagen hydrolysate, for improving joint comfort. International Journal of Food Sciences and Nutrition 2009;60 Suppl 2:99-113.
- Bruyère O, Zegels B, Leonori L. Rabenda V, Janssen A, Bourges C, Reginster JY. Effect of collagen hydrolysate in articular pain: A 6-month randomized, double-blind, placebo controlled study. Complementary Therapies in Medicine 2012;20:124-130.
- Clark KL, Sebastianelli W, Flechsenhar KR, Aukermann DF, Meza F, Millard RL, Deitch JR, Sherbondy PS, Albert A. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Current Medical Research and Opinions 2008;24(5):1485-1496.
- CNF 2023: Canadian Nutrient File (CNF). Nutrition & Healthy Eating, Food and Nutrition, Health Canada. [Accessed 2024 March 8]. Available from: https://food- nutrition.canada.ca/cnf-fce/serving-portion.do?id=1484
- derMarderosian A, Beutler JA, editors. The Review of Natural Products. ?Lysine: Issue date February 2011? St Louis (MO): Facts and Comparisons, Wolters Kluwer Health; Printed in 2008 and Updated to June 2011.
- Eastoe JE. The amino acid composition of mammalian collagen and gelatin. Biochemical Journal 1955;61(4):589-600.
- FCC 7: Food Chemical Codex, Seventh Edition. Rockville (MD): The United States Pharmacopeial Convention, 2010.
- Garrison RH, Somer E. Nutrition Desk Reference, 3rd edition. New Canaan (CT): Keats Publishing, 1995.
- Goldman L, Ausiello D, editors. Cecil Textbook of Medicine, Volume 1, 22nd edition. Philadelphia (PA): Saunders; 2004.
- ICIDH 2008: International Cosmetic Ingredient Dictionary and Handbook, Twelfth Edition, Volume 1. Gottschalck TE, Bailey JE, editors. Washington (DC): The Cosmetic, Toiletry, and Fragrance Association, 2008.
- IOM 2005: Institute of Medicine of the National Academies. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. Food and Nutrition Board, [Accessed 2018 June 5]. Available from: https://www.nal.usda.gov/sites/default/files/fnic_uploads/energy_full_report.pdf
- Jansen GR. Lysine in Human Nutrition. The Journal of Nutrition 1962;76:1-35.
- Moskowitz RW. Role of collagen hydrolysate in bone and joint disease. Seminars in Arthritis and Rheumatism 2000;30(2):87-99.
- NIH 2023: National Institutes of Health. PubChem. Bethesda (MD): National Library of Medicine, US Department of Health & Human Services. [Accessed 2023 October 23]. Available from: https://pubchem.ncbi.nlm.nih.gov/
- Oesser S, Adam M, Babel W, Seifert J. Oral administration of 14C labeled gelatin hydrolysate leads to an accumulation of radioactivity in cartilage of mice (C57/BL). Journal of Nutrition 129(10):1891-5, 1999.
- Ph.Eur. 2023: European Pharmacopoeia. 11th edition. Strasbourg (FR): Directorate for the Quality of Medicines and HealthCare of the Council of Europe (EDQM), 2023.
- Schauss AG, Stenehjem J, Park J, Endres JR, Clewell A. Effect of the novel low molecular weight hydrolyzed chicken sternal cartilage extract, BioCell Collagen, on improving osteoarthritis-related symptoms: a randomized, double-blind, placebo-controlled trial. J Agric Food Chem. 2012 Apr 25;60(16):4096-101
- Schrieber R, Gareis H. Gelatine Handbook: Theory and Industrial Practice. Weinheim: Wiley-VCH. 2007
- Shils ME, Shike M, Ross AC, Caballero B, Cousins RJ, editors. Modern Nutrition in Health and Disease, 10th edition. Philadelphia (PA): Lippincott Williams and Wilkins, 2006.
References reviewed
- American College of Toxicology. Final Report on the Safety Assessment of Hydrolyzed Collagen. International Journal of Toxicology 1985;4:199-221.
- Balian G, Bowes JH. The structure and properties of collagen. In: Ward AG, Courts A, editors. The science and technology of gelatin. London (GB): Academic Press, 1977.
- Barnett ML, Kremer JM, St Clair EW, Clegg DO, Furst D, Weisman M, Fletcher MJ, Chasan-Taber S, Finger E, Morales A, Le CH, Trentham DE. Treatment of rheumatoid arthritis with oral type II collagen. Results of a multicenter, double-blind, placebo-controlled trial. Arthritis and Rheumatism 1998;41(2):290-7.
- Bello AE, Oesser S. Collagen hydrolysate for the treatment of osteoarthritis and other joint disorders: a review of the literature. Current Medical Research and Opinion 2006;22(11):2221-2232.
- Bornstein P, Sage H. Structurally Distinct Collagen Types. Annual Review of Biochemistry 1980;49:957-1003.
- BP 2008: British Pharmacopoeia Commission. 2007. British Pharmacopoeia 2008. Volume I. London (GB): The Stationary Office on behalf of the Medicines and Healthcare products Regulatory Agency (MHRA).
- Clark KL, Sebastianelli W, Flechsenhar KR, Aukermann DF, Meza F, Millard RL, Deitch JR, Sherbondy PS, Albert A. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Current Medical Research and Opinions 2008;24(5):1485-1496.
- Deparle LA, Gupta RC, Canerdy TD, Goad JT, D'Altilio M, Bagchi M, Bagchi D. Efficacy and safety of glycosylated undenatured type-II collagen (UC-II) in therapy of arthritic dogs. Journal of Veterinary Pharmacology and Therapeutics 2005;28(4):385-90.
- Djagny KB, Wang Z, Xu S. Gelatin: A Valuable Protein for Food and Pharmaceutical Industries: Review. Critical Reviews in Food Science and Nutrition 2001;41(6):481-492.
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on Biological Hazards of the European Food Safety Authority on the "Quantitative assessment of the human and animal BSE risk posed by gelatine with respect to residual BSE risk". Question No EFSA-Q-2003-099. Adopted on 18 January 2006. The EFSA Journal 2006:312:1-29.
- FDA 1975: U.S. Food and Drug Administration.
 Database of Select Committee on GRAS Substances (SCOGS)
Reviews.
Gelatin.
[Last Updated: 2006 October 31; Accessed 2012 May 15].
Database of Select Committee on GRAS Substances (SCOGS)
Reviews.
Gelatin.
[Last Updated: 2006 October 31; Accessed 2012 May 15].
- Fini M, Torricelli P, Giavaresi G, Carpi A, Nicolini A, Giardino R. Effect of L-lysine and L-arginine on primary osteoblast cultures from normal and osteopenic rats. Biomedical Pharmacotherapy 2011;55:213-220.
- Fragakis AS, Thomson C. The Health Professional's Guide to Popular Dietary Supplements, 3rd edition. Chicago (IL): American Dietetic Association.
- Fujita T, Ohue M, Fujii Y, Miyauchi A, Takagi Y. The effect of active absorbable algal calcium (AAA Ca) with collagen and other matrix components on back and joint pain and skin impedance. Journal of Bone and Mineral Metabolism 2002;20:298-302.
- Goldman L, Ausiello D, editors. Cecil Textbook of Medicine, Volume 1, 22nd edition. Philadelphia (PA): Saunders; 2004.
- Groff J, Gropper S. Advanced Nutrition and Human Metabolism. 3rd edition. Belmont (CA): Wadsworth/Thomson Learning; 2000.
- Hays NP, Kim H, Wells AM, Kajkenova O, Evans WJ. Effects of whey and fortified collagen hydrolysate protein supplements on nitrogen balance and body composition in older women. Journal of the American Dietetic Association 2009;109(6):1082-7.
- Iwai K, Hasegawa T, Taguchi Y, Morimatsu F, Sato K, Nakamura Y, Higashi A, Kido Y, Nakabo Y, Ohtsuki K, Identification of Food-Derived Collagen Peptides in Human Blood after Oral Ingestion of Gelatin Hydrolysates. Journal of Agricultural and Food Chemistry 2005;53:6531-6536.
- Lee SK, Posthauer ME, Dorner B, Redovian V, Maloney MJ. Pressure ulcer healing with a concentrated, fortified, collagen protein hydrolysate supplement: a randomized controlled trial. Advances in Skin and Wound Care 2006;19(2):92-6.
- Li F, Jia Dongying, Yao K. Amino acid composition and functional properties of collagen polypeptide from Yak (Bos grunniens) bone. Food Science and Technology 2009;42:945-949.
- Martin-Bautista E. Marint-Matillas M, Maring-Lagos JA, Miranda-Leon MT, Muñoz-Torres M, ruiz-Requena E, Rivero M, Quer J, Puigdueta I, Campoy C. A nutritional intervention study with hydrolyzed collagen in pre-pubertal Spanish children: influence on bone modeling biomarkers. Journal of Pediatric Endocrinology and Metabolism 2011;24(3-4):147-53Oesser S, Adam M, Babel W, Seifert J. Oral administration of 14C labeled gelatin hydrolysate leads to an accumulation of radioactivity in cartilage of mice (C57/BL). Journal of Nutrition 1999;129(10):1891-5.
- Merck 2012: The Merck Index Version 14.1. Whitehouse Station (NJ): Merck & Co., Inc. Copyright © 2006, 2012 Merck Sharp &Dohme Corp., a subsidiary of Merck & Co., Inc. [Accessed 2012 May 15]. Available from: http://www.medicinescomplete.com
- Oesser S, Seifert J. Stimulation of type II collagen biosynthesis and secretion in bovine chondrocytes cultured with degraded collagen. Cell and Tissue Research`2003;311(3):393-9.
- Popenoe EA, Aronson RB, Van Slyke DD. Hydroxylysine formation from lysine during collagen biosynthesis. Biochemistry 1966;55:393-397.
- Rowe RC, Sheskey PJ and Owen SC. Handbook of Pharmaceutical Excipients 5th edition. American Pharmacists Association, 2006.
- Schauss AG, Merkel DJ, Glaza SM, Sorenson SR. Acute and subchronic oral toxicity studies in rats of a hydrolyzed chicken sternal cartilage preparation. Food and Chemical Toxicology 2007;45(2):315-21.
- Schrieber R and Gareis H. Gelatine Handbook: Theory and Industrial Practice. Weinheim (DE): Wiley-VCH, 2007.
- Shils ME, Olson JA, Shike M, Ross AC, editors. Modern Nutrition in Health and Disease, 10th edition. Philadelphia (PA): Lippincott Williams and Wilkins, 2006.
- Sieper J, Kary S, Sörensen H, Alten R, Eggens U, Hüge W, Hiepe F, Kühne A, Listing J, Ulbrich N, Braun J, Zink A, Mitchison NA. Oral type II collagen treatment in early rheumatoid arthritis. A double-blind, placebo-controlled, randomized trial. Arthritis and Rheumatism 1996;39(1):41-51.
- Sundell MB, Cavanaugh KL, Pingsheng W, Shintani A, Hakim RM, Ikizler TA. Oral Protein Supplementation Alone Improves Anabolism in a Dose-Dependent Manner in Chronic Hemodialysis Patients. Journal of Renal Nutrition 2009;19(5):412-421.
- Trentham DE, Dynesius-Trentham RA, Orav EJ, Combitchi D, Lorenzo C, Sewell KL, Hafler DA, Weiner HL. Effects of oral administration of type II collagen on rheumatoid arthritis. Science 1993;261(5129):1727-1730.
- Veis A, The macromolecular chemistry of gelatine. New York (NY): Academic Press, 1964.
- Ward AG and Courts A. The Science and Technology of Gelatin. London (GB): Academic Press, 1977.
- Wei W, Zhang LL, Xu JH, Xiao F, Bao CD, Ni LQ, Li XF, Wu YQ, Sun LY, Zhang RH, Sun BL, Xu SQ, Liu S, Zhang W, Shen J, Liu HX, Wang RC. A multicenter, double-blind, randomized, controlled phase III clinical trial of chicken type II collagen in rheumatoid arthritis. Arthritis Res Ther. 2009;11(6):R180.
- Wu J, Fujioka M, Sugimoto K, Mu G, Ishimi Y. Assessment of effectiveness of oral administration of collagen peptide on bone metabolism in growing and mature rats. Journal of Bone and Mineral Metabolism 2004;22(6):547-53.
- Zhang Z, Li G, Shi B. Physiochemical properties of collagen, gelatin and collagen hydrosylate derived from bovine lime split wastes. Journal of the Society of Leather Technologists and Chemists 2006;90(1):32-28.
- Zhao W, Tong T, Wang L, Li PP, Chang Y, Zhang LL, Wei W. Chicken type II collagen induced immune tolerance of mesenteric lymph node lymphocytes by enhancing beta2-adrenergic receptor desensitization in rats with collagen-induced arthritis. International Immunopharmacology 2011;11(1):12-8.
- Zuckley L, Angelopoulou KM, Carpenter MR, McCarthy S, Meredith BA, Kline G, Rowinski M, Smith D, Angelopoulos TJ, Rippe JM. Collagen Hydrolysate Improves Joint Function in Adults with Mild Symptoms of Osteoarthritis of the Knee. Medicine and Science in Sports and Exercise 2004;36(5):S153-S154.