PEPPERMINT - MENTHA X PIPERITA
This monograph is intended to serve as a guide to industry for the preparation of Product Licence Applications (PLAs) and labels for natural health product market authorization. It is not intended to be a comprehensive review of the medicinal ingredient.
Notes
- Text in parentheses is additional optional information which can be included on the label at the applicant's discretion.
- The solidus (/) indicates that the terms and/or statements are synonymous. Either term or statement may be selected by the applicant on the label.
Date
June 27, 2025
Proper name(s), Common name(s), Source information
Peppermint dried leaf
| Proper name(s) | Common name(s) | Source information | ||
|---|---|---|---|---|
| Source material(s) | Part(s) | Preparation(s) | ||
| Mentha x piperita | Peppermint | Mentha x piperita | Leaf | Dry |
References: Proper name: USDA 2024; Common name: Gardner and McGuffin 2013; Source information: ESCOP 2003.
Peppermint essential oil
| Proper name(s) | Common name(s) | Source information | |
|---|---|---|---|
| Source material(s) | Part(s) | ||
| Mentha x piperita | Peppermint essential oil | Mentha x piperita | Leaf |
References: Proper name: USDA 2024; Source information: ESCOP 2003.
Route of Administration
Oral
Dosage Form(s)
This monograph excludes foods or food-like dosage forms as indicated in the Compendium of Monographs Guidance Document.
Acceptable dosage forms for oral use are indicated in the dosage form drop-down list of the web-based Product Licence Application form for Compendial applications.
Use(s) or Purpose(s)
All products
- (Traditionally) used in Herbal Medicine (as a stomachic) to aid digestion (Boon and Smith 2004; Blumenthal et al. 2000; Felter and Lloyd 1983).
- (Traditionally) used in Herbal Medicine (as a carminative) to help relieve flatulent dyspepsia (ESCOP 2003; Hoffmann 2003; Bradley 1992; Felter and Lloyd 1983).
Essential oil
(Traditionally) used in Herbal Medicine to help relieve nausea and vomiting (Boon and Smith 2004; Hoffmann 2003; Blumenthal et al. 2000; Felter and Lloyd 1983).
Notes
- The above uses can be combined on the product label if from the same traditional or non-traditional system of medicine (e.g., Traditionally used in Herbal Medicine to aid in digestion and help relieve flatulent dyspepsia). Does not apply to the essential oil as only one claim is associated with it.
- For multi-ingredient products:
- To prevent the product from being represented as a "traditional medicine", any indicated traditional use claim must refer to the specific medicinal ingredient(s) and recognized traditional system of medicine from which the claim originates when 1) both traditional and modern claims are present or 2) when claims originate from multiple systems of traditional medicine (e.g., Peppermint is traditionally used in Herbal Medicine to aid digestion).
- When ALL of the medicinal ingredients (MIs) in the product are used within the SAME identified system of traditional medicine AND the product makes ONLY traditional claims, listing of MIs in the traditional claim(s) is not required.
Dose(s)
Subpopulation(s)
As specified below.
Quantity(ies)
Methods of preparation: Dry, Powdered, Non-Standardized Extracts (Dry extract*, Tincture, Fluid extract, Decoction, Decoction concentrate, Infusion, Infusion concentrate)
| Subpopulation(s)1,2 | Peppermint dried leaf (g/day) | ||
|---|---|---|---|
| Minimum | Maximum | ||
| Children | |||
| 2-4 years | 0.2 g | 2 g | |
| 5-9 years | 0.3 g | 3 g | |
| 10-11 years | 0.6 g | 6 g | |
| Adolescents | |||
| 12-14 years | 0.6 g | 6 g | |
| 15-17 years | 1.2 g | 12 g | |
| Adults | |||
| 18 years and older | 1.2 g | 12 g | |
1Children and adolescent doses were calculated as a fraction of the adult dose (JC 2008). The use of peppermint leaf in children and adolescents is supported by the following references: McIntyre 2005; Bove 1996.
2Adult dose supported by the following references: Mills and Bone 2005; ESCOP 2003; Blumenthal et al. 2000; Bradley 1992.
*Note: Solvents allowed for the method of preparation “Non-Standardized Extracts (Dry extract)” as part of this monograph are ethanol and/or water only.
Method of preparation: Oil, Essential (water steam distillation)
| Subpopulation(s)1,2 | Peppermint essential oil (μl/day) | ||
|---|---|---|---|
| Minimum | Maximum | ||
| Children | |||
| 2-4 years | 10 μl | 130 μl | |
| 5-9 years | 15 μl | 200 μl | |
| 10-11 years | 30 μl | 400 μl | |
| Adolescents | |||
| 12-14 years | 30 μl | 400 μl | |
| 15-17 years | 60 μl | 800 μl | |
| Adults | |||
| 18 years and older | 60 μl | 800 μl | |
1Children and adolescent doses were calculated as a proportion of the adult dose (JC 2008). The use of peppermint essential oil in children and adolescents is supported by the following references: McIntyre 2005; Bove 1996.
2Adult dose supported by the following references: ESCOP 2003; Blumenthal et al. 2000.
Direction(s) for use
No statement required.
Combination Rule
No permitted combinations between the two medicinal ingredients listed in Tables 1 and 2.
Duration(s) of Use
No statement required.
Risk Information
Caution(s) and warning(s)
All products
- Ask a health care practitioner/health care provider/health care professional/doctor/physician if symptoms persist or worsen.
- Ask a health care practitioner/health care provider/health care professional/doctor/physician before use if you are pregnant or breastfeeding
- Ask a health care practitioner/health care provider/health care professional/doctor/physician before use if you have gallstones or anemia (Brinker 2010; Mills and Bone 2005; Blumenthal et al. 2000).
Essential oil
Ask a health care practitioner/health care provider/health care professional/doctor/physician before use if you have hiatus hernia or gastroesophageal reflux (Brinker 2010; Mills and Bone 2005; ESCOP 2003).
Contraindication(s)
No statement required.
Known adverse reaction(s)
Essential oil
- When using this product you may experience gastroesophageal reflux (Brinker 2010; Mills and Bone 2005; ESCOP 2003).
- Stop use if hypersensitivity/allergy occurs (Mills and Bone 2005; ESCOP 2003).
Non-medicinal ingredients
Must be chosen from the current Natural Health Products Ingredients Database (NHPID) and must meet the limitations outlined in the database.
Storage conditions
Must be established in accordance with the requirements described in the Natural Health Products Regulations.
Specifications
- The finished product specifications must be established in accordance with the requirements described in the Natural and Non-prescription Health Products Directorate (NNHPD) Quality of Natural Health Products Guide.
- The medicinal ingredient must comply with the requirements outlined in the NHPID.
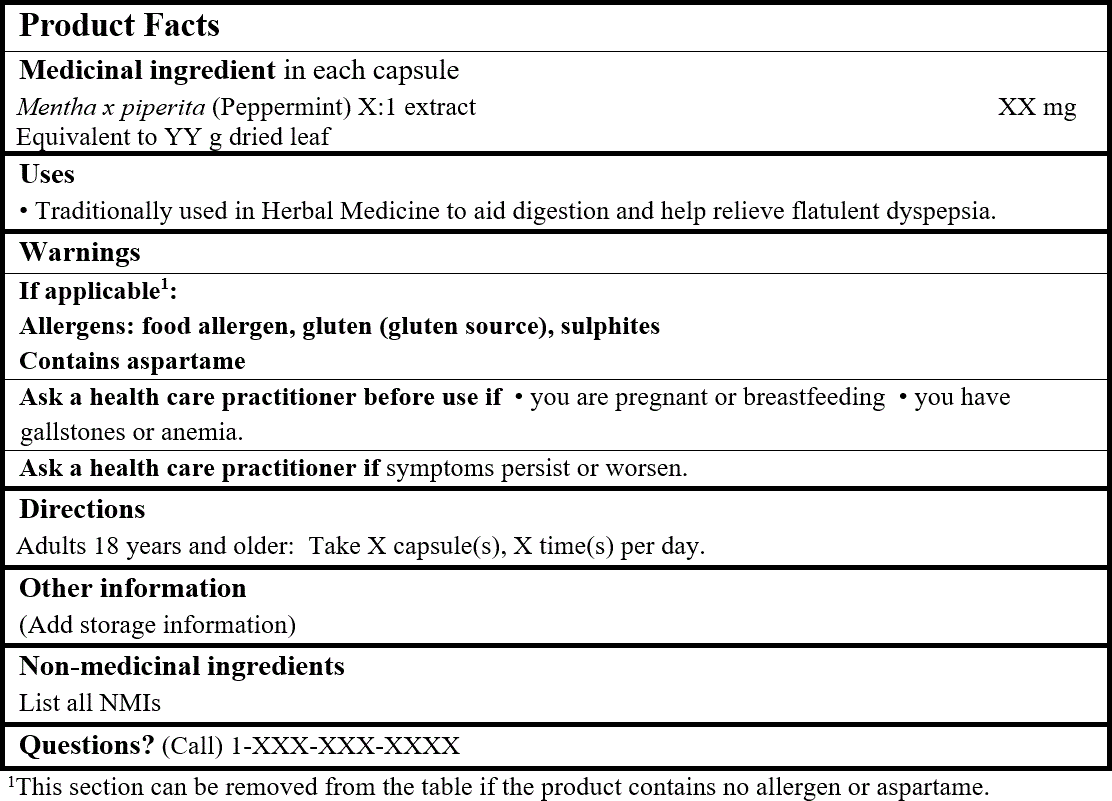
EXAMPLE OF PRODUCT FACTS:
Consult the Guidance Document, Labelling of Natural Health Products for more details.
References Cited
- Blumenthal M, Goldberg A, Brinkmann J, editors. Herbal Medicine: Expanded Commission E Monographs. Boston (MA): Integrative Medicine Communications; 2000.
- Boon H, Smith MJ. The Complete Natural Medicine Guide to the 50 Most Common Medicinal Herbs, 2nd edition. Toronto (ON): Robert Rose Inc; 2004.
- Bove M. An Encyclopedia of Natural Healing for Children and Infants. New Canaan (CT): Keats Publishing, Incorporated; 1996.
- Bradley PR, editor. British Herbal Compendium: A Handbook of Scientific Information on Widely Used Plant Drugs, Volume 1. Bournemouth (UK): British Herbal Medicine Association; 1992.
- Brinker F. Herb Contraindications and Drug Interactions, 4th edition. Sandy (OR): Eclectic Medical Publications; 2010.
- EMA/CHMP 2006: European Medicines Agency: Pre-authorization Evaluation of Medicines for Human Use. Committee for Medicinal Products for Human Use. Reflection Paper: Formulations of choice for the paediatric population. Adopted September 2006.[Accessed 2024 November 29]. Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-formulations-choice-paediatric-population_en.pdf
- ESCOP 2003: ESCOP Monographs: The Scientific Foundation for Herbal Medicinal Products, 2nd edition. Exeter (UK): European Scientific Cooperative on Phytotherapy and Thieme; 2003.
- Felter HW, Lloyd JU. King's American Dispensatory, Volume 2, 18th edition. Sandy (OR): Eclectic Medical Publications; 1983 [Reprint of 1898 original].
- Gardner Z, McGuffin M, editors. American Herbal Products Association's Botanical Safety Handbook. 2nd edition. Boca Ration (FL): Taylor and Francis Group; 2013.
- Giacoia GP, Taylor-Zapata P, Mattison D. Eunice Kennedy Shriver National Institute of Child Health and Human Development Pediatric Formulation Initiative: selected reports from working groups. Clinical Therapeutics 2008; 30(11):2097-2101.
- Hoffmann D. Medical Herbalism. Rochester (VT): Healing Arts Press; 2003.
- JC 2008: Justice Canada. Food and Drug Regulations. (C.01.021). Ottawa (ON): Justice Canada; 2008. [Accessed 2024 November 29] Available from: http://laws.justice.gc.ca/eng/regulations/c.r.c.,_c._870/page-110.html#h-156
- McIntyre A. Herbal Treatment of Children - Western and Ayurvedic Perspectives. Toronto (ON): Elsevier Limited; 2005.
- Mills S, Bone K. The Essential Guide to Herbal Safety. St. Louis (MO): Elsevier Churchill Livingstone; 2005.
- USDA 2024: United States Department of Agriculture, Agricultural Research Service (USDA ARS), Germplasm Resources Information Network (GRIN) - Global. U.S. National Plant Germplasm System. [Accessed 2024 November 14]. Available from: https://npgsweb.ars-grin.gov/gringlobal/taxon/taxonomysearch