MALE GENITAL DESENSITIZERS
Health Products and Food Branch
Date
December 19, 2025
FOREWORD
This monograph describes the requirements necessary to receive market authorization [i.e. a Drug Identification Number (DIN) or a Natural Product Number (NPN)], for male genital desensitizers products. The monograph identifies the permitted medicinal and non-medicinal ingredients, concentrations, indications, directions and conditions of use for these products to be market authorized without the submission to Health Canada of additional evidence. Products which do not meet all of the criteria outlined in this document can apply for market authorization outside of the monograph stream.
Male genital desensitizing products are applied to the penis to help in temporarily slowing the onset of ejaculation.
Applicants are reminded that male genital desensitizers producxts, like other drugs or natural health products, are subject to the Food and Drug Regulations or the Natural Health Products Regulations administered by the Natural and Non-prescription Health Products Directorate (NNHPD). This includes requirements related to labelling, manufacturing and product specifications. Additional information on labels, outside of those specified in the monograph, such as additional directions for use and/or non-therapeutic claims are acceptable as long as they meet the Guidelines for the Nonprescription and Cosmetic Industry Regarding Non-therapeutic Advertising and Labelling Claims, the Guidelines for Consumer Advertising of Health products for Nonprescription drugs, Natural Health Products, Vaccines and Medical Devices, and are not false, misleading or counterintuitive to the use of the product.
The development of this monograph is the result of a thorough review of existing regulations, guidance documents, policies and current practices within Health Canada and other leading regulatory agencies.
Notes
- Text in parentheses is additional optional information which can be included on the label at the applicant's discretion.
- The solidus (/) indicates that either term and/or statement may be selected on the label.
Medicinal ingredient(s)
Male genital desensitizing products are classified as natural health products (NHPs) if they contain the ingredient listed in Table 1 and do not contain the ingredient listed in Table 2. Applicants applying for an NPN can access the appropriate forms and guidance at: https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription.html.
Male genital desensitizing products are classified as non-prescription drugs (NPDs) if they contain the ingredient from Table 2. Applicants applying for a DIN can access the appropriate forms and guidance at: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/applications-submissions/guidance-documents.html.
Proper name(s), Common name(s), Source information
| Proper name(s) | Common name(s) | Source information | Quantity |
|---|---|---|---|
| Source ingredient(s) | |||
| 4-Aminobenzoic acid, ethyl ester | Benzocaine | Benzocaine | 3-7.5%1 |
1Should be prepared in a water-soluble base.
References: Proper name: RSC 2025; Common name: RSC 2025; USP-NF 2024, CTFA 2008; Source information: RSC 2025; CTFA 2008; Quantity: US FDA 2023.
| Proper name(s) | Common name(s) | Quantity |
|---|---|---|
|
Lidocaine | Metered spray containing approximately 10 mg/spray |
References: Proper name: RSC 2025; Common name: RSC 2025; Quantity: US FDA 2023.
Route of administration
Topical
Dosage form(s)
Acceptable dosage forms for NHPs (Products containing benzocaine)
- Aerosol; Aerosol, metered dose; Aerosol, spray; Gel; Liquid; Solution; Spray; Spray (bag-on-valve); Spray, metered dose; Topical liquid; Wipe
Acceptable dosage forms for NPDs (products containing lidocaine)
- Spray, metered dose
Use(s) or Purpose(s)
- For reducing oversensitivity in the male advance of intercourse (US FDA 2023).
- For temporary male genital desensitisation, helping to slow the onset of ejaculation (US FDA 2023).
- Helps in temporarily retarding the onset of ejaculation/temporarily slowing the onset of ejaculation/temporarily prolonging the time until ejaculation (US FDA 2023).
- Helps in the prevention of premature ejaculation (US FDA 2023).
Dose(s)
Subpopulation(s)
Adults 18 years and older
Quantity(ies)
Permitted combinations
Combinations of medicinal ingredients listed in Tables 1 and 2 are not permitted.
Direction(s) for use
Products containing benzocaine
- Apply a small amount to head and shaft of penis 5-10 minutes before intercourse, or use as directed by a health care practitioner/health care provider/health care professional/doctor/physician (US FDA 2023; CPhA 1996).
- Wipe off any excess product before commencing intercourse (CPhA 1996).
- Use smallest amount possible to achieve desired result.
- Wash product off after intercourse (US FDA 2023; CPhA 1996).
Products containing lidocaine
- Apply 3 or more sprays, not to exceed 10, to head and shaft of penis before intercourse, or use as directed by a health care practitioner/health care provider/health care professional/doctor/physician (US FDA 2023).
- Wash product off after intercourse (US FDA 2023).
Spray and aerosol products
- Avoid inhaling or exposing others to spray (USP DI 2006).
Duration(s) of use
Products containing benzocaine
No statement required.
Products containing lidocaine
- Do not use more than 24 sprays in 24 hours.
- Do not use the spray repeatedly for more than 3 months without consulting your doctor.
Risk information
Caution(s) and warning(s)
All products
- For external use only.
- Keep out of reach of children. If swallowed, call a poison control centre or get medical help right away.
- Stop use and ask a health care practitioner/health care provider/health care professional/doctor/physician if this product, used as directed, does not provide relief. Premature ejaculation may be due to a condition requiring medical supervision (US FDA 2023).
Products containing benzocaine
When using this product avoid contact with eyes. If contact occurs, rinse thoroughly with water (US FDA 2023).
Products containing lidocaine
When using this product avoid contact with eyes or nostrils. If contact occurs, rinse thoroughly with water.
Contraindication(s)
All products
- Do not use if your partner is pregnant.
- Do not use if you are elderly.
Products containing benzocaine
Do not use if you or your partner are allergic to benzocaine.
Products containing lidocaine
Do not use if you or your partner are allergic to lidocaine.
Known adverse reaction(s)
All products
- Stop use and ask a health care practitioner/health care provider/health care professional/doctor/physician if you or your partner develop a rash or irritation, such as burning or itching (US FDA 2023).
- Stop use and ask a health care practitioner/health care provider/health care professional/doctor/physician if the following symptoms appear: weakness, confusion, headache, difficulty breathing and/or pale, gray or blue coloured skin. These symptoms may be signs of methemoglobinemia, a rare disorder, which may appear up to 2 hours after use (US FDA 2018; US FDA 2011a,b; HC 2006).
Products containing lidocaine
- Ask a health care practitioner/health care provider/health care professional/doctor/physician if you have or have had liver or kidney problems.
- Ask a health care practitioner/health care provider/health care professional/doctor/physician if you are taking other medications, as high blood level of lidocaine may affect other medicines.
- Stop use and ask a health care practitioner/health care provider/health care professional/doctor/physician if you develop nervousness, dizziness, convulsions, loss of consciousness, any heart or lung symptoms or you feel unwell or have any unusual symptoms.
Non-medicinal ingredients
Ingredients must be chosen from the current Natural Health Products Ingredients Database (NHPID) and must meet the limitations outlined in that database, the Food and Drug Regulations (FDR), and the current Cosmetic Ingredient Hotlist, when relevant.
Storage conditions
Must be established in accordance with the requirements described in the Natural Health Products Regulations.
Specifications
This monograph describes those requirements that are specific to this class of drugs and to NHPs. Any change to the manufacturing process that impacts the safety and efficacy of the ingredients, such as the use of novel technology, requires supporting data and will be reviewed outside the monograph
Products containing Table 1 NHP medicinal ingredients only
The finished product specifications must be established in accordance with the requirements described in the NNHPD Quality of Natural Health Products Guide. The medicinal ingredient must comply with the requirements outlined in the NHPID.
Products containing Table 2 drug medicinal ingredients
Requirements described in the Regulations to the Food and Drugs Act must be met.
EXAMPLE OF PRODUCT FACTS:
Consult the Guidance Document, Labelling of Natural Health Products for more details.
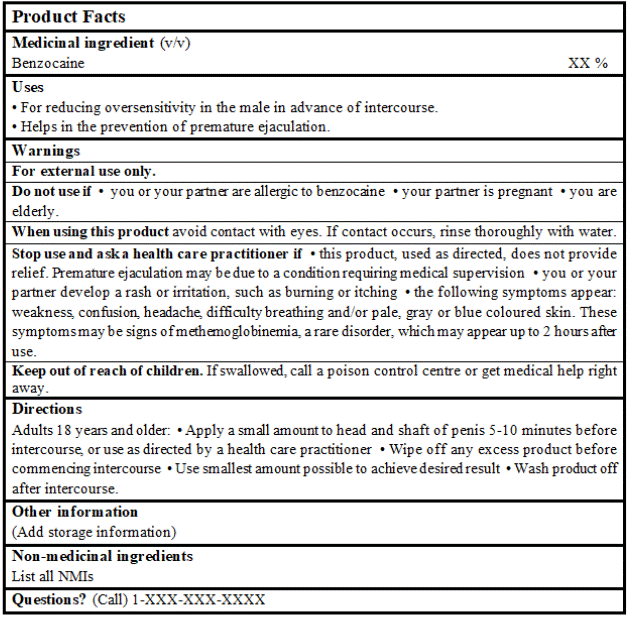
DRUG FACTS TABLE:
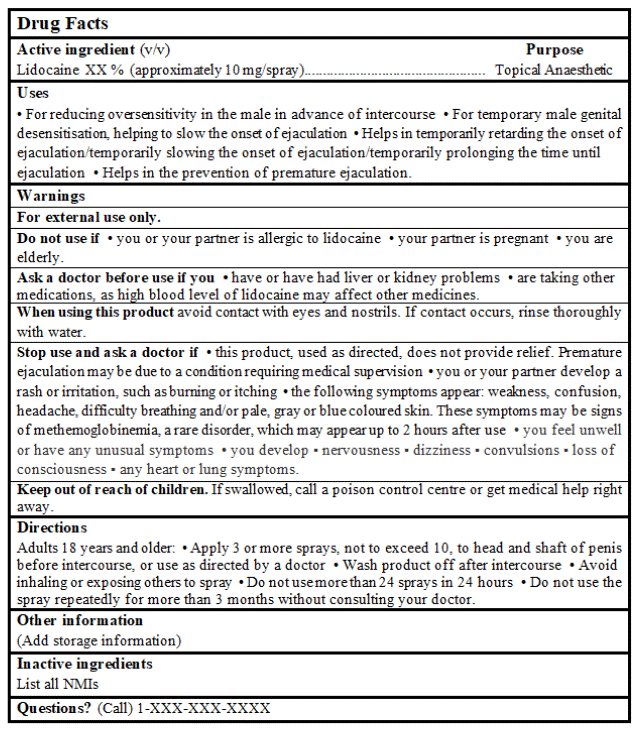
References Cited
- CPhA 1996: Carruthers-Czyzewski P, Gillis C, Letwin D, editors. Nonprescription Drug Reference for Health Professionals. Ottawa (ON): Canadian Pharmaceutical Association; 1996.
- CTFA 2008: Gottschalck TE, Bailey JE, editors. International Cosmetic Ingredient Dictionary and Handbook. 12th edition. Washington (DC): Cosmetic, Toiletry and Fragrance Association; 2008.
- HC 2006: Health Canada advises Canadians of health risks involved with using benzocaine products. [Accessed 2025 November 13]. Available from: https://www.canada.ca/en/news/archive/2006/11/health-canada-advises-canadians-health-risks-involved-using-benzocaine.html
- RSC 2025: Royal Society of Chemistry: The Merck Index Online [Accessed 2025 November 13]. Available from: https://merckindex.rsc.org/
- US FDA 2023. US Department of Health and Human Services, Food and Drug Administration. Over-the-Counter (OTC) Monograph M017: External Analgesic Drug Products for Over-the-Counter Human Use [Accessed 2025 November 05]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTC%20Monograph_M017-External%20Analgesic%20Drug%20Products%20for%20OTC%20Human%20Use%2005.02.2023.pdf
- US FDA 2018. FDA Drug Safety Communication: Risk of serious and potentially fatal blood disorder prompts FDA action on oral over-the-counter benzocaine products used for teething and mouth pain and prescription local anesthetics 05-23-2018. [Accessed 2024 December 16]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/risk-serious-and-potentially-fatal-blood-disorder-prompts-fda-action-oral-over-counter-benzocaine
- US FDA 2011a. FDA continues to receive reports of a rare, but serious and potentially fatal adverse effect with the use of benzocaine sprays for medical procedures 04-07-2011. [Accessed 2024 December 16]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-continues-receive-reports-rare-serious-and-potentially-fatal
- US FDA 2011b. FDA Drug Safety Communication: Reports of a rare, but serious and potentially fatal adverse effect with the use of over-the-counter (OTC) benzocaine gels and liquids applied to the gums or mouth 04-07-2011. [Accessed 2024 December 16]. Available from: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-reports-rare-serious-and-potentially-fatal-adverse-effect-use-over#:~:text=1%20Labels%20of%20marketed%20benzocaine%20products%20currently%20do,products%20out%20of%20reach%20of%20children.%20More%20items.
- USP DI 2006: Drug Information for the Health Care Professional. 26th edition, Volume 1. Greenwood Village (CO): Thomson Micromedex; 2006.
- USP-NF 2024: United States Pharmacopeia and the National Formulary. Rockville (MD): United States Pharmacopeial Convention, Inc.; 2024.
References Reviewed
- Abu-Laban RB, Zed PJ, Purssell RA, Evans KG. Severe methemoglobinemia from topical anesthetic spray: case report, discussion and qualitative systematic review. Canadian Journal of Emergency Medicine 2001;3(1):51-56.
- Ash-Bernal R, Wise R, Wright SM. Acquired methemoglobinemia. A retrospective series. Medicine 2004;83(5):265-273.
- Balicer RD, Kitai E. Methemoglobinemia caused by topical teething preparation: a case report. Scientific World Journal 2004;4:517-520.
- Barker SJ, Tremper KK, Hyatt J. Effects of methemoglobinemia on pulse oximetry and mixed venous oximetry. Anesthesiology 1989;70(1):112-117.
- Benzocaine Topical Products: Sprays, Gels and Liquids - Risk of Methemoglobinemia. [Posted 04/07/2011] U.S. Food and Drug Administration MedWatch The FDA Safety Information and Adverse Event Reporting Program. [Accessed 2025 November 13]. Available from: https://wayback.archive-it.org/7993/20170112165108/http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm250264.htm
- Birchem SK. Benzocane-induced methemoglobinemia during transesophageal echocardiography. Journal of the American Osteopathic Association 2005;105(8):381-384.
- BP 2012: British Pharmacopoeia 2012. London (GB): The Stationary Office on behalf of the Medicines and Healthcare products Regulatory Agency (MHRA); 2011.
- Chung N-Y, Batra R, Itzkevitch M, Borochov D, Balduf M. Severe methemoglobinemia linked to gel-type topical benzocaine use: a case report. The Journal of Emergency Medicine 2010;38(5):601-606.
- Clary B, Skaryak L, Tedder M, Hilton A, Botz G, Harpole D. Methemoglobinemia complicating topical an esthesia during bronchoscopic procedures. Journal of Thoracic and Cardiovascular Surgery 1997;114(2):203-205.
- Dahshan A, Donovan K. Severe methemoglobinemia complicating topical benzocaine use during endoscopy in a toddler: a case report and review of the literature. Pediatrics 2006;117(4):e806- 809.
- Darracq M, Daubert P. A cyanotic toddler. Pediatric Emergency Care 2007;23(3):195-199.
- Dinneen SF, Mohr DN, Fairbanks BF. Methemoglobinemia from topically applied anesthetic spray. Mayo Clinic Proceedings 1994;69(9):886-889.
- Douglas WW, Fairbanks VF. Methemoglobinemia induced by a topical anesthetic spray (cetacaine). Chest 1977;71(5):587-591.
- El-Husseini A, Azarov N. Is threshold for treatment of methemoglobinemia the same for all? A case report and literature review. The American Journal of Emergency Medicine 2010;28(6):748.e5-748.e10.
- Fisher MA, Henry D, Gillam L, Chen C. Toxic methemoglobinemia: A rare but serious complication of transesophageal echocardiography. Canadian Journal of Cardiology 1998;14(9):1157-1160.
- Grauer SE, Giraud GD. Toxic methemoglobinemia after topical anesthesia for transesophageal echocardiography. Journal of the American Society of Echocardiography 1996;9(6):874-876.
- Gray TA, Hawkins S. A PACU crisis: A case study on the development and management of methemoglobinemia. Journal of PeriAnesthesia Nursing 2004;19(4):242-253.
- Guay J. Methemoglobinemia related to local anesthetics: A summary of 242 episodes. Anesthesia & Analgesia 2009;108(3):837-845.
- Health Canada advises Canadians of health risks involved with using benzocaine. About Health Canada, Advisory 2006-115 November 24, 2006. [Accessed 2025 November 13]. Available from: https://www.canada.ca/en/news/archive/2006/11/health-canada-advises-canadians-health-risks-involved-using-benzocaine.html
- Khorasani A, Candido KD, Ghaleb AH, Saatee S, Appavu SK. Canister tip orientation and residual volume have significant impact on the dose of benzocaine delivered by Hurricane spray. Anesthesia & Analgesia 2001;92:379-383.
- Moore TJ, Walsh CS, Cohen MR. Reported adverse event cases of methemoglobinemia associated with benzocaine products. Archives of Internal Medicine 2004 June 14;164:1192-1196.
- Nguyen, ST, Cabrales RE, Bashour CA, Rosenberger TE Jr, Michener JA, Yared J-P, Starr NJ. Benzocaine-induced methemoglobinemia, Anesthesia & Analgesia 2000;90(2):369-371.
- O'Donohue WJ Jr, Moss LM, Angelillo VA. Acute methemoglobinemia induced by topical benzocaine and lidocaine. Archives of Internal Medicine 1980;140(11):1508-1510.
- Olson Ml, McEvoy GK. Methemoglobinemia induced by local anesthetics. American Journal of Hospital Pharmacy 1981;38(1):89-93.
- Orr TM, Orr DL II. Methemoglobinemia secondary to over-the-counter Anbesol. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 1997;84(1):79-81.
- Osterhoudt K. Methemoglobinemia. In: Erickson TB, Ahrens WR, Aks S, Baum C, Ling L, editors. Pediatric Toxicology. 1st edition. New York, NY: McGraw-Hill; 2005.
- Ph.Eur. 2012: European Pharmacopoeia. 7th edition. Strasbourg (FR): Directorate for the Quality of Medicines and HealthCare of the Council of Europe (EDQM); 2012.
- Public Health Advisory: Benzocaine Sprays marketed under different names, including Hurricaine, Topex, and Cetacaine. [Accessed 2025 November 13]. Available from: https://wayback.archive-it.org/7993/20170406202911/https:/www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm124350.htm
- Rodriguez LF, Smolik LM, Abehlik AJ. Benzocaine-induced methemoglobinemia: report of a severe reaction and review of the literature. The Annals of Pharmacotherapy 1994;28(5):643-649.
- Sachdeva R, Pugeda JG, Casale LR, Meizlish JL, Zarich SW. Benzocaine-induced methemoglobinemia. A potentially fatal complication of transesophageal echocardiography. Texas Heart Institute Journal 2003;30(4):308-310.
- So T-Y, Farrington E. Topical benzocaine-induced methemoglobinemia in the pediatric population. Journal of Pediatric Health Care 2008;22(6):335-339.
- Spiller HA, Revolinski DH, Winter ML. Multi-center retrospective evaluation of oral benzocaine exposure in children. Veterinary and Human Toxicology 2000;42(4):228-231.
- Stoelting RK, Miller RD. Basics of Anesthesia. 4th edition. Publisher: Elsevier Science Health Science Division; 2000.
- Tantisattamo E, Suwantarat N, Vierra JR, Evans SJ. Atypical presentations of methemoglobinemia from benzocaine spray. Hawai'i Medical Journal 2011;70(6):125-126.
- Townes PL, Geertsma MA, White MR. Benzocaine-induced methemoglobinemia. American Journal of Diseases of Children 1977;131(6):697-698.
- Tsigrelis C, Weiner L. Methemoglobinemia revisited: an important complication after transesophageal echocardiography. The European Society of Cardiology 2006;7(6):470-472.
- US FDA 2022. US Department of Health and Human Services, Food and Drug Administration. Over-the-Counter (OTC) Monograph M022: Oral Health Care Drug Products for Over-the-Counter Human Use [Accessed 2025 November 05]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/omuf/monographs/OTC%20Monograph_M022-Oral%20Healthcare%20Products%20for%20OTC%20human%20Use%2010.14.2022.pdf
- Vallurupalli M, Das M, Manchanda S. Infection and the risk of topical anesthetic induced clinically significant methemoglobinemia after transesophageal echocardiography. Echocardiography 2010;27(3):318-323.
- Vessely MB, Zitsch RP III. Topical anaesthetic-induced methemoglobinemia: A case report and review of literature. Otolaryngology-Head and Neck Surgery 1993;108(6):763-767.
- VHA Pharmacy Benefits Management Strategic Healthcare Group and the Medical Advisory Panel and the National Center for Patient Safety. A Guidance on the Use of Topical Anesthetics for Naso/Oropharyngeal and Laryngotracheal Procedures; 2006.
- Wong DH, Wilson SE. Avoiding topical anesthesia-induced methemoglobinemia. Obesity Surgery 2005;15(7):1088.
- Wright R, Lewander W, Woolf A. Methemoglobinemia: etiology, pharmacology, and clinical management. Annals of Emergency Medicine 1999;34(5):646-656.