ATHLETE'S FOOT TREATMENTS
Help on accessing alternative formats, such as Portable Document Format (PDF), Microsoft Word and PowerPoint (PPT) files, can be obtained in the alternate format help section.
Date
2018-12-07
FOREWORD
This monograph is intended to replace the existing Athlete's Foot Treatments Monograph of September 11, 1995. This monograph describes the requirements necessary to receive market authorization [i.e. a Drug Identification Number (DIN) or a Natural Product Number (NPN)], for athlete's foot treatment products. The monograph identifies the permitted medicinal and non-medicinal ingredients, concentrations, indications, directions and conditions of use for these products to be market authorized without the submission to Health Canada of additional evidence. It also may contain the test methods recommended to be used to comply with the requirements of this monograph. Products which do not meet the criteria outlined in this document can apply for market authorization outside of the monograph stream.
Applicants are reminded that athlete's foot treatments products, like other non-prescription drugs or natural health products, are subject to the Food and Drug Regulations or the Natural Health Products Regulations administered by the Natural and Non-prescription Health Products Directorate (NNHPD). This includes requirements related to labelling, manufacturing and product specifications. Additional information on labels, outside of those specified in the monograph, such as additional directions for use and/or non-therapeutic claims are acceptable as long as they meet the Guidelines for the Nonprescription and Cosmetic Industry Regarding Non-therapeutic Advertising and Labelling Claims, the Guidelines for Consumer Advertising of Health products for Nonprescription drugs, Natural Health Products, Vaccines and Medical Devices, and are not false, misleading or counterintuitive to the use of the product.
The development of this monograph is the result of a thorough review of existing regulations, guidance documents, policies and current practices within Health Canada and other leading regulatory agencies.
Note:
The solidus (/) indicates that the terms and/or the statements are synonymous. Either term or statement may be selected by the applicant. Text in parentheses is additional optional information which can be included on the Product Licence Application form and label at the applicant's discretion.
Medicinal Ingredient(s)
Athlete's foot treatment products are classified as natural health products (NHPs) if they contain only ingredients from Table 1. Applicants applying for an NPN can access the appropriate forms and guidance at: https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription.html.
Athlete's foot treatment products are classified as non-prescription drugs if they contain at least one ingredient from Table 2. Applicants applying for a DIN can access the appropriate forms and guidance at: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/applications-submissions/guidance-documents.html.
Route(s) of Administration
Topical
Dosage Form(s)
Acceptable dosage forms for the age category listed in this monograph and specified route of administration are indicated in the Compendium of Monographs Guidance Document.
Use(s) or Purpose(s)
Self-Care Framework Category I Uses or Purposes:
For all products, the following statements may be made:
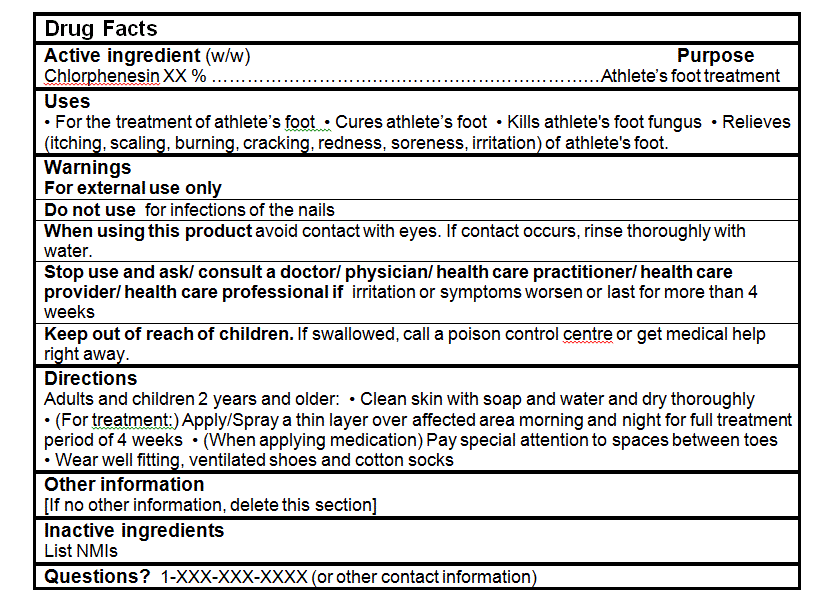
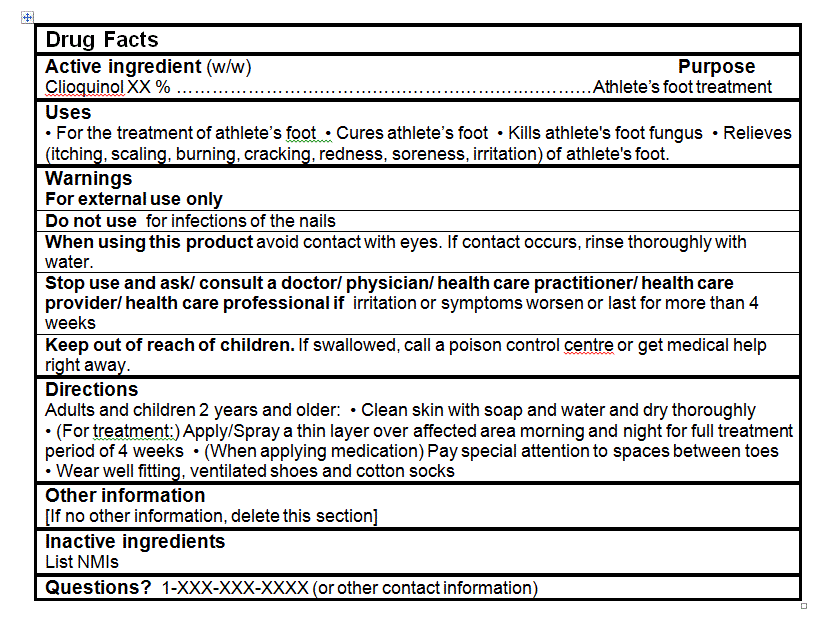
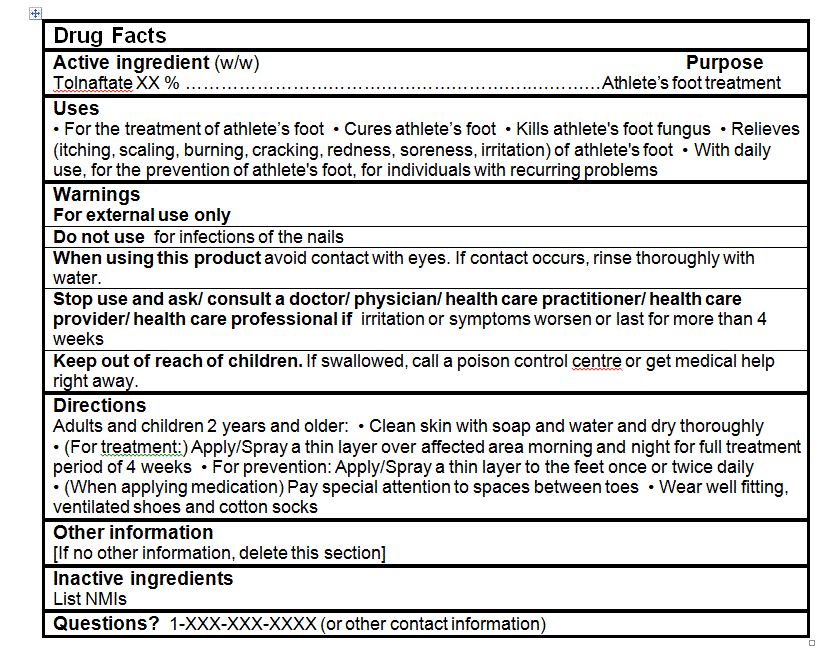
- For the treatment of athlete's foot
- Cures athlete's foot
- Kills athlete's foot fungus
- Relieves (itching, scaling, burning, cracking, redness, soreness, irritation) of athlete's foot.
For products containing tolnaftate, the following statement may be made:
- With daily use, for the prevention of athlete's foot, for individuals with recurring problems.
Dose(s)
Subpopulation(s):
Children 2 to 11 years, Adolescents 12 to 17 years, Adults 18 years and olderQuantity
See Tables 1 and 2 above.Permitted combinations:
Two or more of the following ingredients from Table 1 may be combined, provided that the combined ingredients provide a total undecylenate concentration of 10 - 25%:- undecylenic acid
- calcium undecylenate
- copper undecylenate
- zinc undecylenate
Directions for use:
For all products:
- Clean skin with soap and water and dry thoroughly
- (For treatment:) Apply/Spray a thin layer over affected area morning and night for full treatment period of 4 weeks
- (When applying medication) Pay special attention to spaces between toes
- Wear well fitting, ventilated shoes and cotton socks
For products containing tolnaftate with a claim for the prevention of athlete's foot:
- For prevention: Apply/Spray a thin layer to the feet once or twice daily
Duration(s) of use:
No statement requiredRisk Information
Caution(s) and Warning(s)
For all products:
- For external use only
- When using this product avoid contact with eyes. If contact occurs, rinse thoroughly with water.
- Stop use and ask/ consult a doctor/ physician/ health care practitioner/ health care provider/ health care professional if irritation or symptoms worsen or last for more than 4 weeks.
- Keep out of reach of children. If swallowed, call a poison control centre or get medical help right away.
Contraindication(s):
For all products:- Do not use for infections of the nail
Known Adverse Reaction(s):
No statement requiredNon Medicinal Ingredients
Ingredients must be chosen from the current Natural Health Products Ingredients Database (NHPID) and must meet the limitations outlined in that database, the Food and Drug Regulations (FDR), and the current Cosmetic Ingredient Hotlist, when relevant.
Storage condition(s)
No statement required
Specifications
This monograph describes those requirements that are specific to this class of non-prescription drugs and to natural health products (NHPs). Any change to the manufacturing process that impacts the safety and efficacy of the ingredients, such as the use of novel technology (e.g. nanotechnology), requires supporting data and will be reviewed outside the monograph.
For products containing Table 1 NHP medicinal ingredients only:
The finished product specifications must be established in accordance with the requirements described in the NNHPD Quality of Natural Health Products Guide. The medicinal ingredient must comply with the requirements outlined in the NHPID.
For products containing Table 2 non-prescription drug medicinal ingredients:
Requirements described in the Regulations to the Food and Drugs Act must be met.
DRUG FACTS TABLES (Format Optional for Self-Care Category I)

References
- Compendium of therapeutics for minor ailments (CTMA). Power, B, et al., editors. Canadian Pharmacists Association ; 2016.
- Compendium of products for minor ailments (CPMA). Power, B, et al., editors. Canadian Pharmacists Association ; 2016.
- Drug Facts and Comparisons, 2016. Facts and Comparisons Division, Wolters Kluwer; 2017 edition (Oct. 28 2016)
- Drugdex, Micromedex Inc. 2.0
- FDA 1993: USA Department of Health and Human Services: Food and Drug Administration. 21 CFR Part 333. Topical antimicrobial Drug for Over-the-Counter Human Use, Part C Topical Antifungal Drug Products Final Monograph; [Accessed August 28, 2018]. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=333
- Goodman and Gilman's The Pharmacological Basis of Therapeutics, 13th Edition 2017 Brunton L., Knollmann B., Hilal-Dandan R. McGraw-Hill Education.
- Krinsky DL, Ferreri SP, Hemstreet B, Hume AL, Newton GD, Rollins CJ, Tietze KJ. Handbook of Nonprescription Drugs: An interactive approach for Self-Care, 19th edition. Washington (DC): American Pharmaceutical Association; 2017.
- Remington's Pharmaceutical Science, 13th Edition, 1996, Philadelphia College of Pharmaceutical Sciences
- Sweetman SC, editor. Martindale: The Complete Drug Reference, 39th edition. Grayslake (IL): Pharmaceutical Press; 2017.